A joint technical article by Rehm Thermal Systems and ViscoTec Pumpen- u. Dosiertechnik
In modern medical technology, maximum precision in the application of adhesives, coatings, or potting compounds is vital. Especially for medical devices in the highest risk class III, such as implants, the safety and reliability of the end product depend significantly on exact dosing. Microdispensing plays a key role wherever the finest structures must be joined or sealed without impairing the product’s function or flexibility.
Microdispensing as a key factor in medical technology
A clear example of the need for precise dispensing is the deep brain stimulator (DBS). A DBS system delivers targeted electrical impulses to the brain via extremely fine electrodes and thus makes a major contribution to the treatment of conditions such as Parkinson’s disease or therapy-resistant epilepsy. Selective adhesive bonds ensure that every single contact point and every block segment is reliably sealed before the next process step. This primary step prevents components from shifting during potting or gaps from opening that would allow the potting compound to penetrate uncontrollably. A secondary barrier is then created by potting that completely encapsulates the components. This produces a homogeneous protective layer over all components to ensure long-term biocompatibility as well as high vibration and shock resistance of the implant. Because body fluids tend to migrate into the gap between the primary and secondary barriers, in some cases it is advisable to apply an adhesion promoter in between to provide even greater protection and a long-term stable seal.
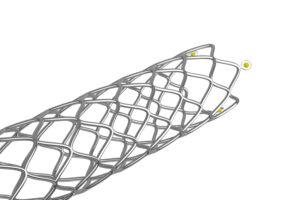
Fig. 1: Stent with radiopaque markers – filled with minimal amounts of adhesive (Image source: ViscoTec)
Exact adhesive application is also essential in the manufacture of stents. In particular, stents made from materials with low radiopacity, such as nitinol, are difficult to see in X-ray images. To enable precise positioning in the body nonetheless, tiny radiopaque markers are applied. For this purpose, gaps of only 15–30 µm are filled with minimal quantities of adhesive. These micro-doses preserve the stent’s mobility, maintain its flexibility, and at the same time ensure long-term biocompatibility.
Fig. 2: Size comparison of a micro adhesive dot with a 2.5 mm dispensing syringe tip (Image source: ViscoTec)
Another typical field of application for Microdispensing is catheters – highly flexible, soft systems used in many minimally invasive procedures. Here, a wide variety of components, such as connectors, must be joined within tight tolerances. Adhesives are dispensed as tiny, precisely placed dots so that they hold reliably while neither impairing the catheter’s flexibility nor restricting flow in its lumens. Overdosing could lead to constrictions, blockages, or even particle detachment, while underdosing would create mechanical weak points.
Whether pacemakers, stents, or catheters – in all products it is crucial that the adhesive is applied exactly at the intended location and in the correct quantity. Only controlled application with minimal tolerances reliably minimizes risks, ensures full component functionality, and meets the high requirements for safety, quality, and long-term stability.
Technological challenges and solutions
Microdispensing places high demands on dispensing system precision, since even the smallest deviations can significantly affect the result. The axis accuracy of the dispensing platform must be extremely precise and repeatable. Among the greatest challenges, however, are material variations caused, for example, by temperature differences, entrapped air, or manufacturer-related viscosity deviations. Time/pressure dispensing systems reach their limits here because their dosing result depends heavily on the fill level of the pressurized cartridge: a full, half-full, or nearly empty cartridge leads to different application volumes, since air compresses very differently than liquid. In medical technology, exactly reproducible dispensing is essential, because inaccurate quantities not only lead to scrap and rework, but in the worst case can pose a direct risk to the patient.

Fig. 3: The preeflow eco-PEN XS was developed specifically for the smallest dispensing volumes. (Image source: ViscoTec)
In addition to its own applicators, Rehm Thermal Systems has been relying for some time on ViscoTec’s precise dispensing technology. A key advantage is the progressive cavity (eccentric screw) technology installed in the dispensers, which is integrated, among other things, into Protecto systems. With volumetric conveying via a rotating screw in an elastic stator, the material is transported uniformly and without pulsation. This achieves very high repeatability and makes the process largely independent of viscosity fluctuations. In contrast to time/pressure-based systems, conveying is purely volumetric – the screw displaces exactly the same volume per revolution, regardless of the cartridge fill level. Another decisive advantage: material variations such as temperature changes or fillers have little effect, because the screw can convey even highly viscous or filled media gently and at a constant delivery rate. Even with sensitive media, the material structure is not damaged, and there is no air entrapment or phase separation.
To avoid air entrapment between the substrate and the dispensed material, other factors also play an important role. Not only correct programming of material application – such as targeted filling of capillaries – but also subsequent drying and curing is a decisive factor. With an exactly coordinated oven profile, the materials cure completely and form a smooth, homogeneous end result. Depending on the chemical composition of the materials used, Rehm RDS drying systems are configured individually. Different physical principles of heat transfer, such as IR or UV radiation, are used to make the drying processes as efficient as possible.
Quality assurance through maintenance and calibration
In validated medical-technology production processes, regular maintenance and calibration are crucial to ensure permanently accurate and reproducible dispensing results. A clean applicator is a particular focus – an indispensable basis for flawless application. ViscoTec’s precise dispensers also excel here: the progressive-cavity screw can rotate both forwards and backwards and offers a programmable retraction for a defined string break without dripping. This prevents unwanted material discharge after the dispensing step.

Fig. 4: Precise material application on complex geometries with an integrated 3D height sensor (Image source: Rehm)
In addition, a cleaning tape is available for Rehm Protecto systems that automatically cleans the dispense needles before each application process and thus ensures consistently high dispensing quality. The dispensing program is loaded under software control and contains all relevant parameters such as material type, nozzle diameter, dispensing volume, and flow behavior. Consistent material output is additionally monitored via high-precision load cells so that deviations can be detected and prevented at an early stage. To reliably determine location and position even for soft or complex-shaped medical products, the systems feature automatic needle measurement, which checks the applicators at defined intervals and automatically readjusts them if necessary. An integrated fiducial camera detects alignment marks, corrects the zero point, and ensures precise operation even if the assembly is not positioned accurately in the workpiece carrier. In addition, a 3D height sensor automatically compensates for any warpage or instability – for example, in very thin substrates.
Flexibility and process reliability in regulated environments
In medical technology, flexibility plays a central role – depending on the application. In prototype construction or with frequently changing product variants, a typical low-volume–high-mix scenario applies: small batch sizes, different geometries, and constantly new requirements for materials and processes. Here, it is essential that dispensing systems can be adjusted to new applications without conversion and “on the fly.”
Flexibility does not only mean quick program changes, but also the ability to process a wide range of materials in parallel – from highly viscous silicones and low-viscosity acrylates to filled epoxy resins. Modern systems must therefore be able to automatically load and reliably implement different nozzle types, needle lengths, application patterns, and process parameters. Integration into higher-level software solutions also plays a central role: material parameters and dispensing paths are managed centrally and automatically loaded depending on the product, minimizing changeover times and increasing system availability.
At the same time, one must bear in mind that in regulated high-volume production – such as for active implants like pacemakers – flexibility is inherently limited. Here, each production line is validated for a specific product; changes to the process, geometry, or material are only permissible with extensive revalidation. In these cases, process stability takes priority over flexibility, and dispensing systems are usually tailored precisely to one product.
Overall, flexibility remains a decisive factor – especially wherever innovative products, customer-specific variants, or agile development cycles are required. It is not only economically beneficial, but also a prerequisite for producing modern medical technology efficiently and safely.
Automation, regulation, and traceability
Regulatory requirements such as the Medical Device Regulation (EU MDR) are increasingly confronting medical-technology manufacturers with complex tasks. In addition to process reliability, compliance with safety and hygiene requirements plays a central role. As a result, automation is becoming ever more important. This can be seen in developments in the pharmaceutical sector, where EU Annex 1 already stipulated for 2023 that operator intervention must be minimized as far as possible. It can be assumed that this trend will increasingly be reflected in medical technology as well. In addition, systems often need to be suitable for cleanroom use – for example, Rehm Protecto systems for medical technology are designed for an ISO 7 environment.
Material compatibility is also particularly important in all areas that come into contact with the dispensing medium. This applies above all to metallic surfaces through which the materials are conveyed. To ensure biocompatibility, aluminum surfaces are often replaced by stainless-steel variants. UV-curing acrylates, biocompatible epoxy resins, silicones, and other specially approved medical dispensing materials are frequently used. When selecting the right adhesive or potting compound, a key factor is whether it is a long-term implant that remains in the body for years or a temporary system. For long-term implants, the material used must be permanently biocompatible – especially if the bond is in direct contact with tissue or bodily fluids. If the bond, on the other hand, is located inside a biocompatible potting compound or is completely enclosed in a welded titanium housing, the material itself does not necessarily have to be biocompatible. In such cases, aspects such as adhesion properties and curing time tend to be the main focus.

Fig. 5: Customized line solution – including coating/dispensing, drying, handling, and traceability (Image source: Rehm)
Data monitoring and analysis are an integral part of automation in modern medical-technology production and are understood not only as a quality-assurance measure, but also as a regulatory necessity. In a validated environment, production processes must be traceable for many years. If a problem occurs – for example, if individual products are faulty at the customer – it is not sufficient to roughly narrow down a production period. Systems must be able to evaluate exactly which serial numbers are affected, which process parameters were present during dispensing, and under which conditions the product was manufactured. This enables targeted recall actions without unnecessarily removing intact products from the market. This is made possible by consistent traceability with individually configurable interfaces and continuous data acquisition. Temperatures, material batches, serial numbers, and process histories can be captured and synchronized horizontally between machines and reported vertically to higher-level MES systems. In addition, material and process interlocks provide extra safety: before each start, the system checks whether the currently loaded program actually matches the intended product and material. If anything does not match, the process is not released and is automatically locked. These measures achieve seamless traceability and enable immediate response to process deviations – making a decisive contribution to patient safety in medical technology.
From development to series production – “Design to Automation
A particular challenge lies in scaling prototypes up to series production. New products go through lengthy development and validation phases, and only once all processes are qualified may they be transferred to series production. To shorten these timeframes, it is crucial to consider future automation already during product development – an approach known as “Design to Automation.”
At its in-house Technology Center, Rehm – together with ViscoTec – supports customers and interested parties as early as the development phase by carrying out practical trials using original products. Under realistic conditions, optimum process parameters for later series processes can be identified and potential challenges addressed at an early stage. This close collaboration upfront makes it possible to significantly accelerate the transition from development to production and to avoid later adjustments or revalidation.
Precise, gentle, and reliable: Microdispensing with ViscoTec and preeflow
Volumetric dispensers such as the systems from ViscoTec and preeflow enable continuous, material-sensitive dispensing of one- or two-component fluids and pastes – regardless of viscosity. Especially in medical technology, where smooth and sterile surfaces are essential, the retractable mechanism ensures a clean string break without dripping. With dispensing accuracy of up to ±1%, adhesive quantities in the microliter range can be applied precisely and reproducibly, allowing even the smallest components such as catheters, balloons, or connectors to be bonded reliably. The process provides a high level of process reliability. Depending on requirements, the systems are used as stand-alone devices, in automated systems, or integrated into control and switch cabinets. In particular, compact preeflow dispensers are suitable both for manual dispensing tasks in the lab or production and for automated manufacturing of medical-technology components. Their robustness has already proven itself in the automotive and electronics industries and has been specifically adapted to medical standards.

Fig. 6: 1K pressure head vipro-HEAD during bonding of a pacemaker (Image source: ViscoTec)
The new vipro-HEAD medical grade pressure heads were developed specifically for applications where safety, material compatibility, and patient-specific care are the focus. All product-contact parts are made of stainless steel or plastic-/elastomer-based materials with FDA certification; an autoclavable variant up to 121 °C is also available.
With the preeflow eco-PEN XS 180, a particularly powerful micro-dispenser is available. It was designed for ultra-small dispensing volumes and is considered the most precise volumetric system in its class today. Its compact design makes integration into tight pitch dimensions easier and enables parallel operation of multiple units. Progressive cavity technology ensures extremely gentle, low-shear conveying – an important advantage for sensitive or highly filled adhesives as used in high-end medical devices.
Conclusion
Microdispensing is now a decisive building block in the production of modern medical devices. It combines precision, material expertise, and process reliability in the smallest possible space. With innovative technologies such as progressive-cavity dispensing, intelligent calibration and monitoring systems, and consistent traceability, companies such as Rehm Thermal Systems and ViscoTec create the basis for reliable, safe, and cost-effective manufacturing processes in medical technology.



